NanoEx Interactive Regenerating Wound Dressing Matrix
Advanced Regenerating Dermal Matrix for Soft Tissue Repair.
NanoEx® Interactive Regenerating Wound Dressing Matrix is an advanced wound care solution designed to support tissue repair through a combination of structural support, optimized wound environment management, ECM-mimetic composition and antibacterial protection.
CE-marked since 05/2021 and manufactured under an ISO 13485-certified quality management system, NanoEx is developed for integration into modern wound care protocols where effective interaction with the wound bed and support of physiological healing processes are essential.
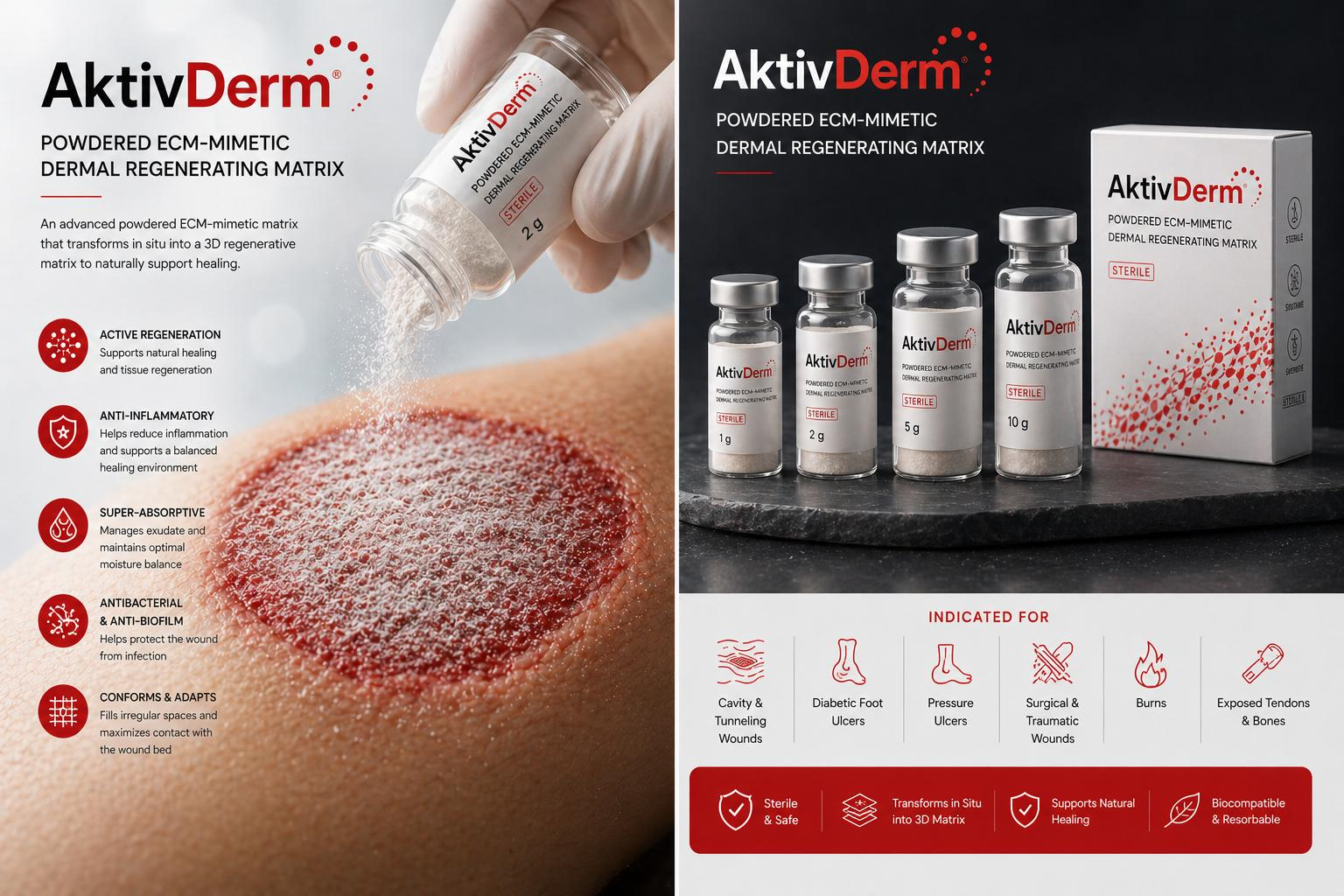
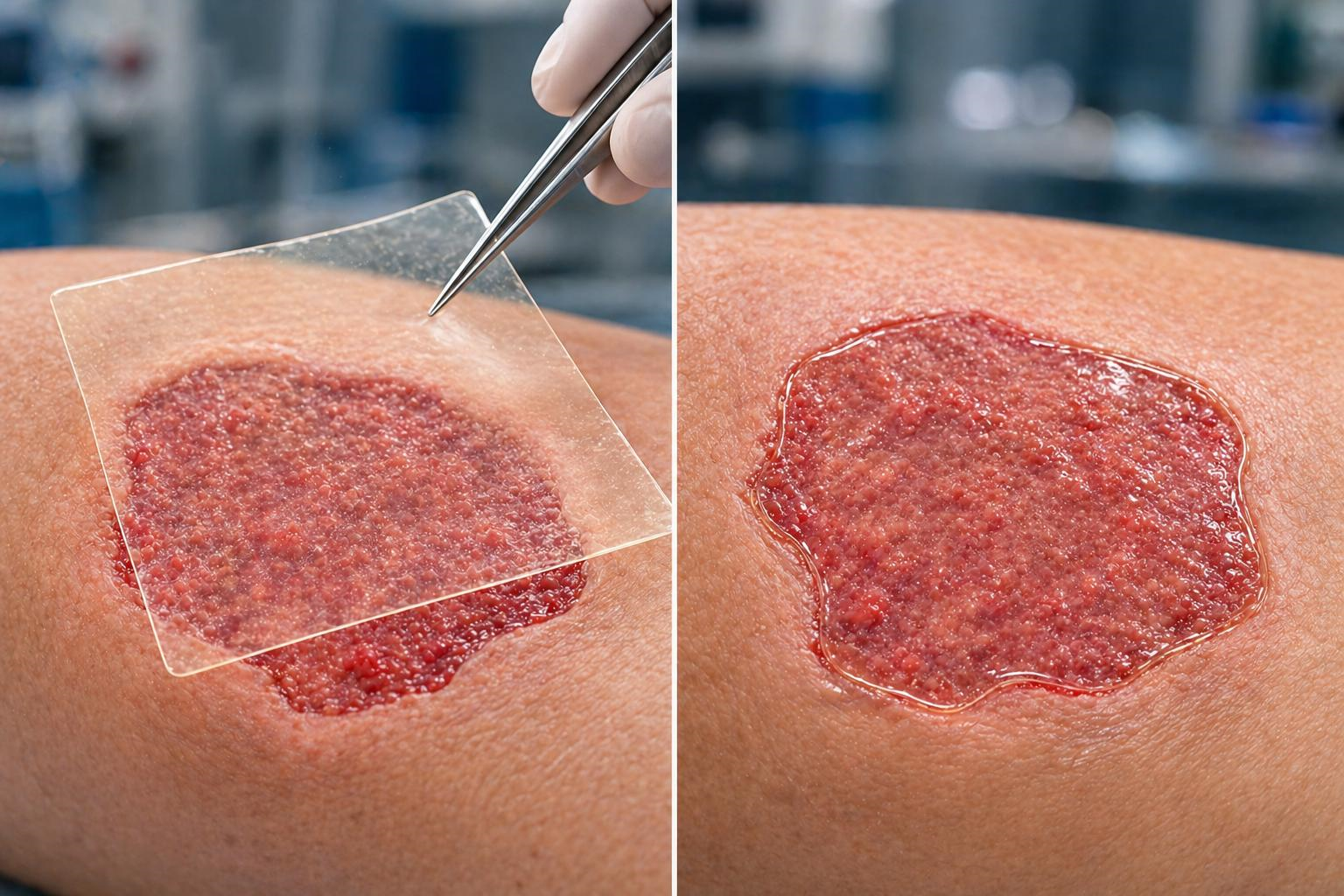
NanoEx is provided as a pre-formed, sterile dermal matrix intended for direct application to the wound bed, offering immediate coverage and structural support.
Mechanism of Action
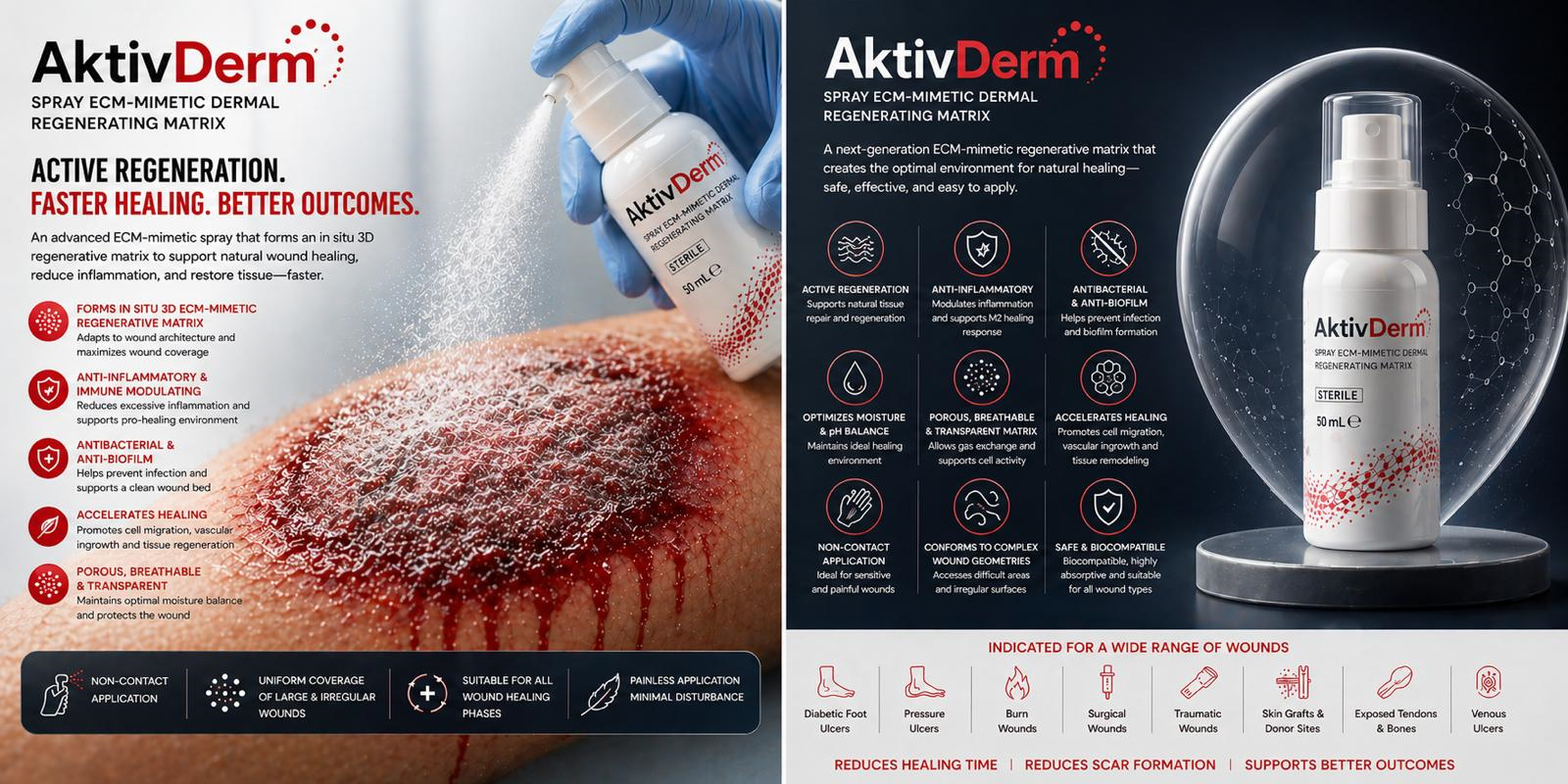
Upon application, NanoEx functions as a pre-structured three-dimensional matrix that interacts with the wound environment to establish a stable, porous, and moisture-permeable scaffold.
- Conforms closely to the wound surface, including irregular geometries
- Maintains consistent contact with the wound bed
- Supports an optimized moist wound environment when used with appropriate secondary dressings
- Provides a physical ECM-mimetic scaffold that supports cellular migration, proliferation and tissue formation
- Contributes to modulation of the wound microenvironment toward a regenerative profile
- Supports fibroblast and endothelial cell activity associated with tissue repair
- Encourages formation of vascularized granulation tissue
- Provides antibacterial and anti-biofilm functionality within the wound environment
- Facilitates the progression of the wound healing process toward tissue repair
The dermal matrix is designed to mimic key structural characteristics of the extracellular matrix (ECM), creating a supportive microenvironment conducive to granulation tissue formation and progressive tissue remodeling. It helps facilitate transition from prolonged inflammatory conditions toward healing progression.
Clinical Advantages and Properties
- Advanced wound care dermal matrix with interactive functionality
- Provides immediate structural scaffold upon application
- Biocompatible and designed for tissue compatibility
- Moisture-vapor permeable
- Porous, breathable, and transparent structure
- Supports pH regulation within the wound environment
- Conforms to wound surface and complex anatomies
- Flexible and adaptable to wound contours
- Supports use across different stages of wound healing
- Supports efficient wound healing progression
- Demonstrated contribution to improved healing outcomes, including reduced healing time and minimized scar formation when used appropriately
- Suitable for application over exposed tendons and bones
- Can be used over joints without restricting mobility
- Painless and non-traumatic application
Indications
NanoEx is indicated for the management of:
- Partial and full-thickness wounds
- Diabetic foot ulcers
- Pressure ulcers
- Burn wounds
- Traumatic wounds
- Surgical wounds
- Infected and non-infected wounds
- Skin grafts and donor sites
- Wounds with exposed tendons and bones
- Cavity wounds
- Dry wounds and wounds with low to moderate exudation
Unlike absorbent wound dressings, NanoEx is non-exudate absorbing and is intended to function as a regenerative scaffold rather than a fluid management solution. Appropriate superabsorbent secondary dressings or combination with Negative Pressure Wound Therapy (NPWT) should be used to manage exudate when necessary.
The matrix is biocompatible, moisture-permeable, and engineered to support an optimal regenerative microenvironment.
Packaging
- 10 x 10 cm, cuttable to the shape of the wound.
- Provided sterile in primary packaging and sealed within a sterile secondary pouch.